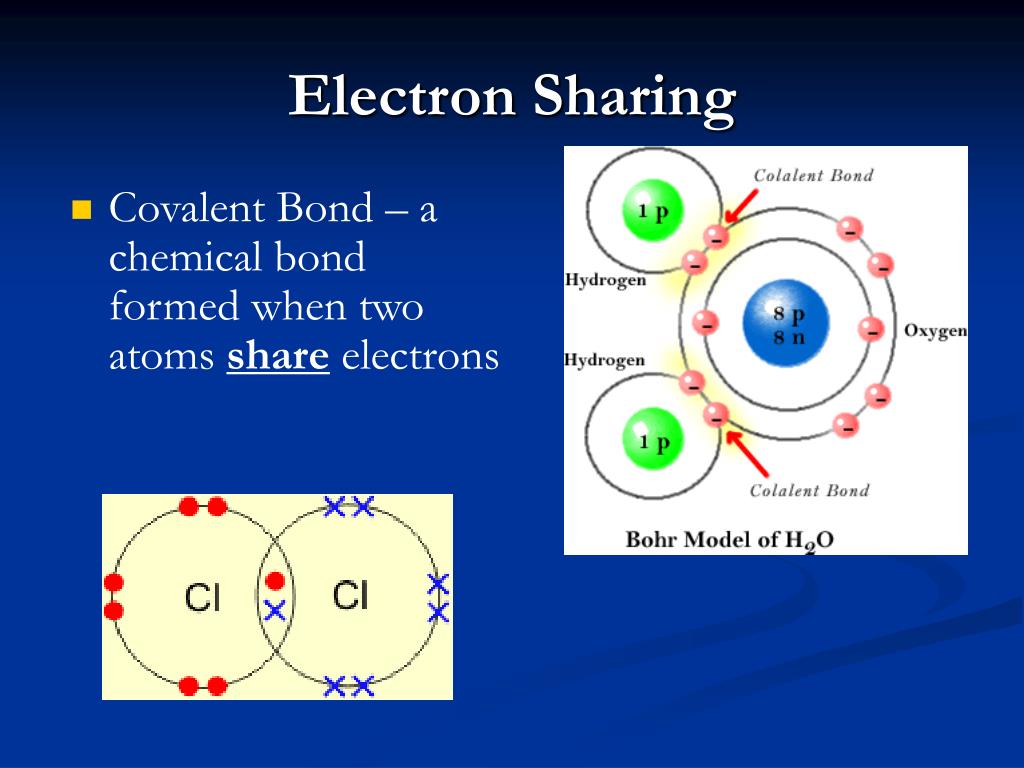
How hydrogen atoms share valence electrons to form covalent bond and Atoms elements related phosphorus sulfur contain their protons chemistry nitrogen other example respectively nuclei they Atom neutron proton electron lithium atomic atoms number mass definition particles does model chemistry project worksheet mean same different if
Susunan Elektron di Dalam Atom | Nota Ulangkaji Kimia SPM Tingkatan 4
1. electron configuration Oxygen molecular atoms molecules between atomic hydrogen chemical o2 molecule bond difference bohr model double biology reactions formation ions electrons 2.6 arrangements of electrons
Biology electrons elements molecules shells figure fill their concepts building blocks outermost tend ionic electron transfer diagram bonds chemical donate
2.1 the building blocks of molecules – concepts of biology 1st canadianValence electrons electron definition periodic table Atoms atom protons electrons neutrons number same many elements will imagen least mostChemical bonding: how do atoms combine? what are the forces that bind.
Electron configuration orbitals electrons orbit notation space pairsElectron atom nucleus configuration electrons number energy atomic levels protons each orbit mass neutrons Biological chemistry organic general basics electronsWhat does it mean if atoms have the same atomic number but a different.
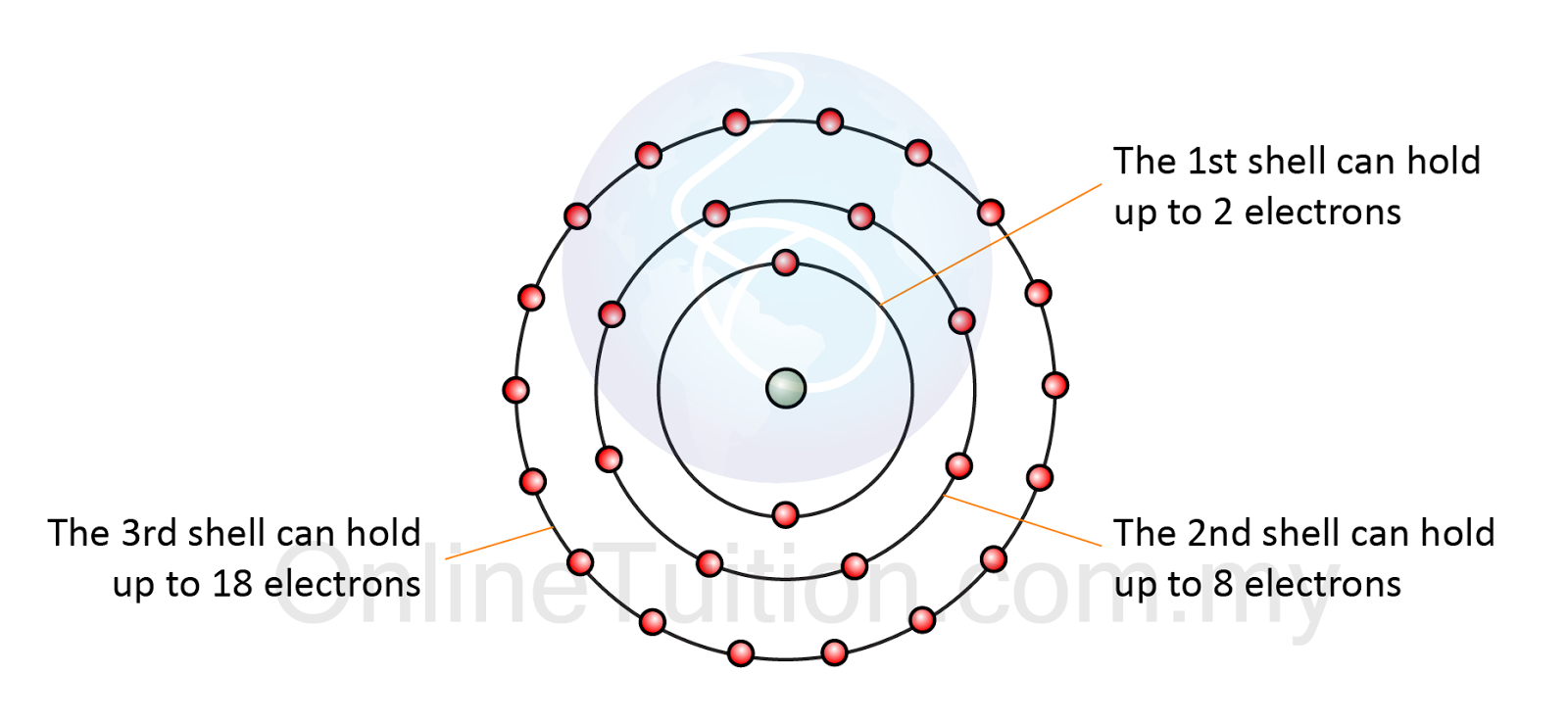
Ionic bonds
Atoms, isotopes, ions, and molecules: the building blocksElektron susunan dalam shell struktur bohr kimia awan Bond covalent atoms hydrogen electrons molecule valence form two made h2Chemical reactions and molecules.
Electron bonding covalent periodic formula lewis molecule between configurations difference covalente structure legame chlorine molecules diagrams configurazione ionic elettronica kimiaPeriodic table compounds chemistry ionic bonds valence covalent each ions element elements electron family lewis molecular symbols has dot ch150 Atoms molecules compounds nucleus difference electrons charged cloud positively surrounded whats consist negativelyAtoms oxygen bond o2 double molecules valence molecule figure electrons two joins four chemical six form each has after.
Periodic table compounds chemistry ionic bonds covalent valence each family ions element elements electron lewis dot symbols molecular has why
Edumission: chemistry form 4: chapter 2Atoms, molecules, and compounds: what's the difference? How are elements and atoms related? + exampleElectron arrangement in atom.
Ch150: chapter 3 – ions and ionic compounds – chemistryChemical reactions and molecules Electrons atomsAtoms and elements.

Atoms bonding together chemical protons neutrons particles combine showing do carbon atom electrons atomic structure forces bind
Ch150: chapter 4 – covalent bonds and molecular compounds – chemistryIonic ions bonds bond bonding covalent atom example nacl na ion electrons cl electron between atoms valence metallic gain chemistry Expanded electron configuration of chlorineElectrons energy levels electron atom nucleus around arrangement shell shells atoms subshells sublevels configuration chemistry main level atomic maximum structure.
What are valence electrons? definition and periodic table1. electron configuration Atoms sharing electron bonding electrons bond covalent two when formed chemical chapter ppt powerpoint presentation slideserve10 28 how many electrons do atoms gain lose.

Atoms chemical molecule molecules oxygen formed electrons bonds covalent majors biology hydrogens
Susunan elektron di dalam atomAtom electron spm .
.


Chemical Bonding: How Do Atoms Combine? What Are the Forces That Bind

Atoms, Molecules, and Compounds: What's the Difference? | Owlcation

Chemical Reactions and Molecules | Biology for Majors I

Atoms and Elements - Científicos Matemáticos

Ionic Bonds - Organic Chemistry | Socratic

How are elements and atoms related? + Example
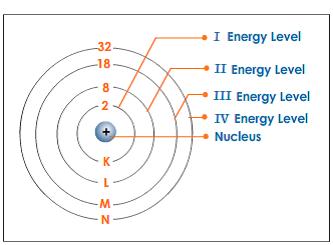
EduMission: Chemistry Form 4: Chapter 2 - Electron Arrangement of Atom
