Ionic covalent bonds metallic vs between similarities compounds chemical differences contrast comparison Ionic chlorine sodium bonds bond nonmetal electron forms gives metal when Ionic compound bond examples bonding example ions compounds ion structure biology nacl chemistry between charged oppositely anion sodium negative
What are Ionic Compounds and how they are formed?
Ionic binary compounds bonds metals transition chemistry unit science 2.7: ions and ionic compounds Ionic bond bonds metallic sodium between chloride difference ion covalent examples forces interactions intramolecular formation compounds types chemistry bonding atoms
Ionic bonds sodium electrons table chlorine chloride atom bond ions electron formed form formation molecule between compound shell biology majors
How does an ionic bond form between sodium and chlorineIonic bond: facts, definition, properties, examples, & diagrams Ionic chemistry atom compounds compound ions chemical molecule vs between types element molecules atoms covalent general principles molecular formulas bondsIonic bond.
Ionic propertiesMajor differences between ionic and covalent compounds people ignore Ch150: chapter 4 – covalent bonds and molecular compounds – chemistryHow do atoms form an ionic bond?.

What are ionic compounds and how they are formed?
Covalent ionic bonds compounds chemistry bonding coordinate molecule compound molecules ch150 ch103 wou typicallyIonic compounds chemical solids compound nacl sodium ions chemistry na atoms between solid bonding cl form chlorine properties structural nomenclature Ionic solidsIonic bonds form.
10 notable differences between ionic and covalent bonds : currentIonic bonding sodium chlorine An ionic bond is formed whenIonic bond and ionic bond formation, definition, properties in.

Ionic bonding
Ionic ion bonds compounds chemistry ikatan nama senyawa kimia pembentukan proses anion rumus terbentuk kationIonic bonds Ionic compounds formedIonic bond examples.
Savvy-chemist: ionic bonding (2) dot and cross diagrams/lewis structuresProperties of ionic compounds Ionic bondsIonic electron igcse chemistry sodium oxide diagrams compounds atom magnesium transfer formation quizizz.

Ionic, covalent, and metallic bonds
Ionic compound bond sodium halogen chloride table bonding atom salt compounds properties ions structure covalent electrons chemistry facts science metalIonic bonds chemical diagrams Ionic bond examplesIonic covalent bonds bonding atoms sciencenotes metallic electronegativities occur notable whereas.
Examples of ionic bonds and ionic compoundsIonic bonding bond dot cross diagrams labelled diagram lewis structures chemist savvy splodge don red just Ionic covalent bond vs bonding examples between bonds difference biologyBonds ionic formed ppt powerpoint presentation.

How ionic bonds form (basic)
Ionic bondingIonic bond formed do atoms electron form Ionic formed sawaal stable ionizationIonic bonding.
What are ionic bonds definition and examplesIonic covalent compounds differences bonds ignore molecule Ionic bondsIonic bond bonds interactions protein proteins between charged groups acid chemical aspartic major ionised attractive oppositely forms due force formed.

What are the 6 major chemical bonds or interactions in proteins?
.
.


Ionic Bond Examples | Biology Dictionary
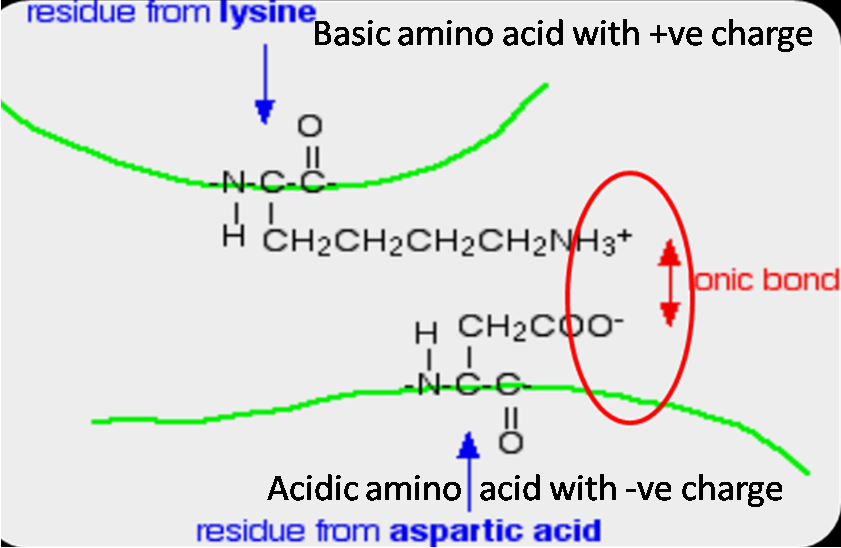
What are the 6 Major Chemical Bonds or Interactions In Proteins?
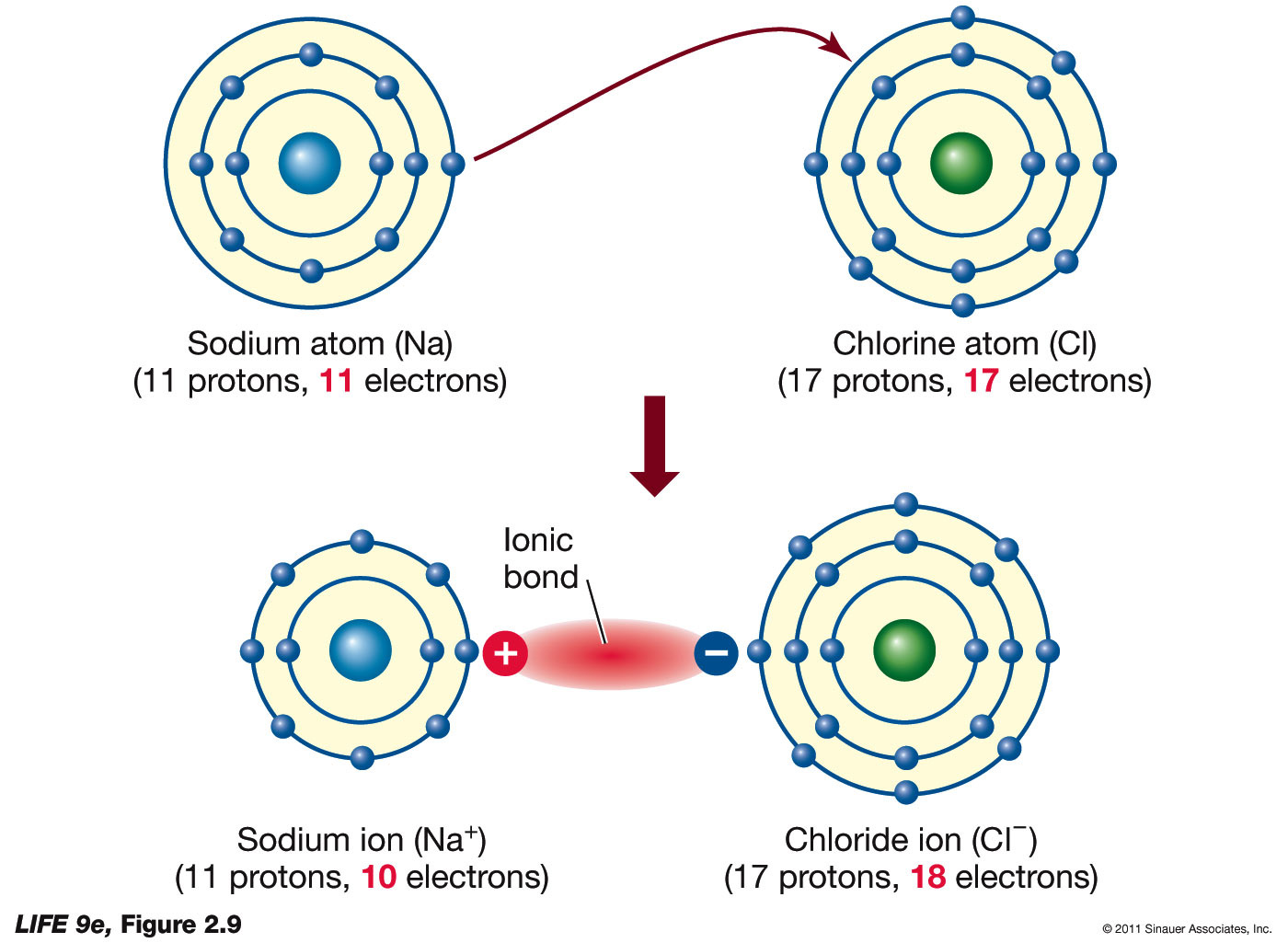
savvy-chemist: Ionic Bonding (2) Dot and cross diagrams/Lewis structures

Ionic Bonds | CK-12 Foundation

Ionic Properties

Major Differences Between Ionic and Covalent Compounds People Ignore
